|
Monitoring your Body's PH levels
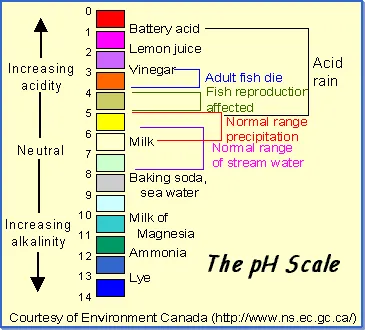
pH: What does it mean? pH is the abbreviation for potential
hydrogen. The pH of any solution is the measure of its hydrogen-ion
concentration. The higher the pH reading, the more alkaline and
oxygen rich the fluid is. The lower the pH reading, the more
acidic and oxygen deprived the fluid is. The pH range is from
0 to 14, with 7.0 being neutral. Anything above 7.0 is alkaline,
anything below 7.0 is considered acidic.
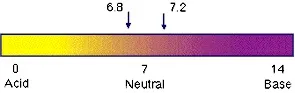
Human blood stays in a very narrow pH range right around (
7.35 - 7.45 ). Below or above this range means symptoms and disease.
If blood pH moves to much below 6.8 or above 7.8, cells stop
functioning and the patient dies. The ideal pH for blood is 7.4
A healthy blood pH without cancer has acid + alkaline balance
almost equal. Actually a healthy body is slightly alkaline measuring
approximately 7.4. This ideal blood 7.4 pH measurement means
it is just slightly more alkaline than acid.
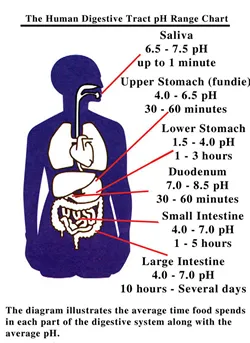 |
The pH in the human digestive tract varies greatly (see
Human Digestive Tract pH Range Chart on the left side). The
pH of saliva is usually between 6.5 - 7.5. After we chew
and swallow food it then enters the fundic or upper portion
of the stomach which has a pH between 4.0 - 6.5. This is
where "predigestion" occurs while the lower portion
of the stomach is secreting hydrochloric acid (HCI) and pepsin
until it reaches a pH between 1.5 - 4.0. After the food mixes
with these juices it then enters the duodenum (small intestine)
where the pH changes to 7.0 - 8.5. This is where 90% of the
absorption of nutrients is taken in by the body while the
waste products are passed out through the colon (pH 4.0 -
7.0).
If you have a health problem, most likely you are acidic. Research
shows that unless the body's pH level is slightly alkaline, the
body cannot heal itself. So, no matter what type of modality
you choose to use to take care of your health problem, it won't
be as effective until the pH level is up. If your body's pH is
not balanced, you cannot effectively assimilate vitamins, minerals
and food supplements. Your body pH affects everything. |
Total healing of chronic illness
only takes place when and if the blood is restored to
a normal, slightly alkaline pH. |
An Imbalance In the
body’s pH may lead to serious health concerns, including:
Hormone concerns
Cardiovascular weakness
Weight gain/loss
Bladder and kidney concerns.
Immune deficiency
Acceleration of free radical damage.
Structural system weakness, including brittle bones, hip fractures
and joint discomfort
Stressed liver function
Low energy
Slow digestion and elimination
Yeast/fungal overgrowth.
Tumor growth
The body has to have a balanced pH like most living things on
earth or it does not function correctly. The alkaline level is
very important because research has already proven that disease
cannot survive in an alkaline state and yet they thrive in an
acidic environment.
 |
The truth is everyone has different nutrient requirements,
but we all share one thing in common - we need to have alkaline
blood to stay healthy. |
An acidic balance will: decrease the body's ability
to absorb minerals and other nutrients, decrease the energy production
in the cells, decrease it's ability to repair damaged cells,
decrease it's ability to detoxify heavy metals, make tumor cells
thrive, and make it more susceptible to fatigue and illness.
An acidic pH can occur from, an acid
forming diet, emotional stress, toxic overload,
and/or immune reactions or any process that deprives the
cells of oxygen and other nutrients. The body will try to
compensate for acidic pH by using alkaline minerals. If the
diet does not contain enough minerals to compensate, a build
up of acids in the cells will occur.
There are two factors that are ALWAYS present with cancer no
matter what else may be present. Those two factors are Acid pH
and Lack of Oxygen. Can we manipulate those two factors that
always have to be present for cancer to develop and by doing
so may help reverse the cancer? If so, we need to learn how to
manipulate those two factors.
Cancer needs an acid and low oxygen environment to survive and
flourish within. Terminal cancer patients are around 1000 times
more acidic than normal healthy people. The vast majority of
terminal cancer patients possess a very low body pH. Why?
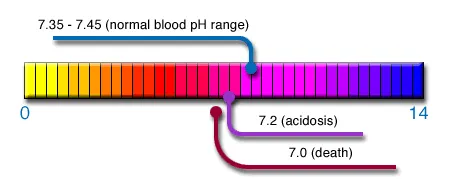
In the absence of oxygen, glucose undergoes fermentation to
lactic acid. This causes the pH of the cell to drop from between
7.3 to 7.2 down to 7 and later to 6.5 in more advanced stages
of cancer and in metastases the pH drops to 6.0 and even 5.7
or lower. Our bodies simply can not fight disease if our body
pH is not properly balanced.
 |
Shown here are four representations chemists
use for molecular oxygen. In colored molecular models, oxygen
is traditionally shown in red.
|
The normal human cell has a lot of molecular oxygen
and a slightly alkaline pH. The cancer cell has an acid pH and
lack of oxygen. Cancer cells cannot survive in an oxygen rich
environment. At a pH slightly above 7.4 cancer cells become dormant
and at pH 8.5 cancer cells will die while healthy cells will
live. Again, the higher the pH reading, the more alkaline and
oxygen rich the fluid is. Cancer and all diseases hate oxygen
/ pH balance. The proper alkalinity pH of the blood ( 7.35 -
7.45 ) is critical for the overall health of the body. In other
words... alkalize or die.
Baking
Soda and pH Levels
Remember that the pH number is an exponent number of 10; therefore,
a small difference in pH translates to a big difference in the
number of oxygen or OH-ions. A difference of 1 in a pH value
means ten times the difference in the number of OH-ions, a difference
of 2 means one hundred times the difference in the number of
OH-ions. In other words, blood with a pH value of 7.45 contains
64.9% more oxygen than blood with a pH value of 7.30.
|
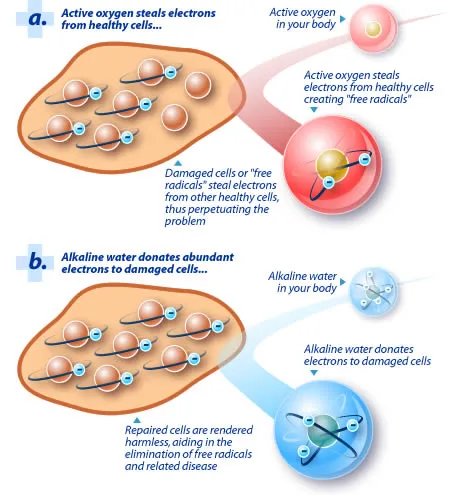
Blood cells have a negative charge on
the outside and a positive
charge inside; this is what keeps them healthy
and far apart from each other. However, when your body
is over-acidic, the acid strips your blood of it's negative
charge. Your blood cells no longer have the same repelling
force and clump together like this: |
Fact: If your body's pH is not balanced, you cannot
effectively assimilate vitamins, minerals and food supplements.
Also, mucus on the small intestine can block your body from vitamin
and mineral absorption.
Oxidation Reduction Potential (ORP)

When your tap water arrives at your home,
it will usually be a little over neutral in
pH ~ around 7.5.
It will, however, be robbed of its electrical
negative charge and will almost always have
a 'positive' ORP .
This means it has no antioxidant abilities
and cannot assist your body in the fight against
oxygen free radicals. On the contrary, it actually
robs you of energy as you drink it.
The measurement of this positive or negative
charge is expressed in millivolts, so an ORP
reading on our ORP meter of tap water will most
likely depending on your area show around +350
millivolts. After it has been through Ionizing
Water filters it will most likely show anything
from -150 to -650mv (depending on the amount
of soluble minerals in the water and on the original
pH of the water).
ORP is a "potential" energy that is
stored and ready to be put to work. An analogous
way to look at ORP would be to consider pressure.
When you blow up a balloon you use energy that
creates force. As long as the balloon remains
closed, this force is a “stored potential
energy” in this case measured as air pressure.
When released, this “potential” energy
becomes kinetic energy.More
Ideal
for all ORP testing,
FEATURES
• Measures Oxidation Reduction Potential (Redox) and Temperature
• Auto-ranging calibration with digital fine tuning
• Includes storage solution in a sponge embedded in a clear cap
• Waterproof housing (IP-67 rating)
• Simultaneous temperature display
• Digital Calibration (push button)
• Auto-off function, data-hold function and low-battery indicator.
• Display: large and easy-to-read LCD screen includes simultaneous
temperature reading.
• Replaceable Sensor (model SP-O2)
• Factory Calibrated: The ORP-200 meter is factory calibrated to
+92 mV The meter can be easily recalibrated with digital calibration using the
push buttons, rather than a screwdriver.
• Includes a cap, batteries, and lanyard
Specifications
ORP Range: -999 to +1000 mV
Temperature Range: 0-80°C; 32-176°F
Resolution: 1 mV; Temperature resolution is 0.1°C/F
Accuracy: 0.5% (+/-2mV)
Calibration: Digital calibration by push button.
Housing: IP-67 Waterproof (submersible; floats)
Power source: 3 x 1.5V button cell batteries (LR44 or equivalent; included)
Dimensions: 18.5 x 3.4 x 3.4 cm (7.3 x 1.3 x 1.3 inches)
Weight: 96.4 g (3.4 oz) |

|
 |
The
American Marine ORP / REDOX Calibration
Fluid
400 mV
|
|
How To
Test Your pH Level...
Salivary pH Test: While generally more acidic than blood, salivary
pH mirrors the blood ( if not around meals ) and is also a fairly
good indicator of health. It tells us what the body retains.
Salivary pH is a fair indicator of the health of the extracellular
fluids and their alkaline mineral reserves.
Optimal pH for saliva is 6.4 to 6.8. Spit onto to your Portable
PH Meter on arising before anything is put into the mouth. A
reading lower than 6.4 is indicative of insufficient alkaline
reserves. After eating, the saliva pH should rise to 7.8 or higher.
Unless this occurs, the body has alkaline mineral deficiencies
( mainly Calcium and Magnesium ) and will not assimilate food
very well. To deviate from ideal salivary pH for an extended
time invites illness.
Acidosis, an extended time in the acid pH state, can result
in rheumatoid arthritis, diabetes, lupus, tuberculosis, osteoporosis,
high blood pressure, most cancers and many more. If salivary
pH stays too low, the diet should focus on fruit, vegetables
and mineral water as well as remove strong acidifiers such as
sodas, whole wheat and red meat.
Urinary pH Test: The pH of the urine indicates how the body
is working to maintain the proper pH of the blood. The urine
reveals the alkaline building (anabolic) and acid tearing down
(catabolic) cycles. The pH of urine indicates the efforts of
the body via the kidneys, adrenals, lungs and gonads to regulate
pH through the buffer salts and hormones. Urine can provide a
fairly accurate picture of body chemistry, because the kidneys
filter out the buffer salts of pH regulation and provide values
based on what the body is eliminating. Urine pH can vary from
around 4.5 to 9.0 for its extremes, but the ideal range is 5.8
to 6.8.
Are you Dehydrated
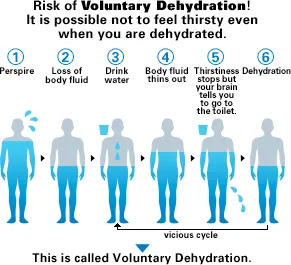
Individuals' daily water requirements can
vary due to many factors - including amount
of exercise performed, total body weight, total
lean muscle weight, and the temperature and
climate of your locale. Many people wait until
they are thirsty before reaching for a bottle
of water. However, thirst is a very unreliable
measure of the water needs for our bodies.
The body can lose up to 2 or more cups of water
before thirst strikes. Not getting enough water
may result in a faster heart rate, thicker-than-normal
blood, lowered endurance capacities, and muscle
cramps. Drinking water in regular intervals
throughout the day is an important habit to
develop. More |
Foods considered to be alkaline-forming and thus helpful to
people with consistently acid pH include: almonds, aloe vera,
apples, apricots, bee pollen, buckwheat, cabbage, cantaloupe,
celery, carrots, cucumbers, dairy products except hard cheese,
dates, dulse, poached eggs, figs, grapefruit, honey, lettuce,
millet, parsley, raisins, peaches, fresh red potatoes, pineapple,
soy products, sprouted seeds, cooked spinach, turnip tops, wakame
miso soup, azuki beans, rice, mineral water. More alkaline-forming
foods...Click Here
People who remain too acid often display symptoms such as: anxiety,
diarrhea, dilated pupils, extroverted behavior, fatigue in early
morning, headaches, hyperactivity, hypersexuality, insomnia,
nervousness, rapid heartbeat, restless legs, shortness of breath,
strong appetite, high blood pressure, warm dry hands and feet.
Nano Energy Flask Portable Water Alkaline pH Enhancer Increases the pH level
in your drinking water
More Information Click here
Balancing the pH is a major step toward well-being and greater
health. Acidosis ( overly acidic body ) is the primary indicator
of Calcium Deficiency Disease. Scientists have discovered that
the body fluids of healthy people are alkaline ( high pH ) whereas
the body fluids of sick people are acidic ( low pH ).
MONITOR YOUR PH DAILY
 |
High Quality
Ideal for all pH testing,
water purification applications, wastewater regulation,
aquaculture, hydroponics, colloidal silver, labs & scientific testing, pools & spas, .
FEATURES
� Measures pH and Temperature
� Auto-ranging three point calibration with digital
fine tuning
� Includes storage solution in a sponge embedded in
a clear cap
� Waterproof housing (IP-67 rating)
� Simultaneous temperature display
� Measurement Range: 0-14 pH
� Digital automatic calibration (one point), with digital
fine tuning
� Automatic Temperature Compensation (ATC)
� Auto-off function, data-hold function and low-battery
indicator
� Display: large and easy-to-read LCD screen includes
simultaneous temperature reading
� Factory Calibrated: The PH-200 meter is three-point
checked and factory calibrated to pH 7.0. It can be
re-calibrated to any point in its range with digital
calibration using the push buttons.
� Includes a cap, batteries, lanyard, and pH 7.0 buffer
|
|

